|
Related Topics
|
|
Category: Catalent (CTLT)
Date: 30 December 2020 Stock price of Catalent (CTLT): $103.56 In this article we take a more detailed look at Catalent (CTLT) a leading global provider of advanced delivery technologies and development and manufacturing solutions for drugs; protein, cell, and gene therapy biologics; and consumer health products
|
|
Catalent employs over 11,000 people, including over 1,400 scientists, at more than 30 facilities across five continents, and in fiscal 2017 generated over $2 billion in annual revenue. "
More About Catalent
We are the leading global provider of advanced delivery technologies and development and manufacturing solutions for drugs; protein, cell, and gene therapy biologics; and consumer health products. Our oral, injectable, cell and gene therapy, and respiratory delivery technologies address the full diversity of the pharmaceutical industry, including small molecules; protein, cell, and gene therapy biologics; and consumer health products. Through our extensive capabilities and deep expertise in product development, we can help our customers take products to market faster, including nearly half of new drug products approved by the U.S. Food and Drug Administration (the “FDA”) in the last decade. Our advanced delivery technology platforms, which include those in our Softgel and Oral Technologies, Biologics, and Oral and Specialty Delivery segments, our proven formulation, manufacturing, and regulatory expertise, and our broad and deep intellectual property enable our customers to develop more products and better treatments for patients and consumers. Across both development and delivery, our commitment to reliably supply our customers’ and their patients’ needs is the foundation for the value we provide; annually, we produce more than 74 billion doses for nearly 7,000 customer products, or approximately 1 in every 20 doses of such products taken each year by patients and consumers around the world.
Catalent employs over 11,000 people, including over 1,400 scientists, at more than 30 facilities across five continents, and in fiscal 2017 generated over $2 billion in annual revenue.
We believe that, through our investments in growth-enabling capacity and capabilities, our ongoing focus on operational and quality excellence, the sales of existing and introduction of new customer products, our innovation activities and patents, and our entry into new markets, we will continue to benefit from attractive and differentiated margins and realize the growth potential from these areas. We continue to invest in our sales and marketing activities, leading to growth in the number of active development programs for our customers. This has further enhanced our extensive, long-duration relationships and long-term contracts with a broad and diverse range of industry-leading customers. In fiscal 2020, we conducted business with 86 of the top 100 branded drug marketers, 24 of the top 25 generics marketers, 23 of the top 25 biologics marketers, and 22 of the top 25 consumer health marketers globally. Selected key customers include Pfizer, Johnson & Johnson, GlaxoSmithKline, Novartis, Roche, and Teva.
We have many long-standing relationships with our customers, particularly in advanced delivery technologies, where we tend to follow a prescription molecule through its lifecycle, from the development and launch of the original brand prescription, to potential generics or an over-the-counter switch. A prescription pharmaceutical product relationship with an innovator will often last many years, in several cases, two decades or more, extending from pre-clinical development through the end of the product’s life cycle. We serve customers who require innovative product development, superior quality, advanced manufacturing, and skilled technical services to support their development and marketed product needs. Our broad and diverse range of technologies closely integrates with our customers’ molecules and other active ingredients to yield final formulations and dose forms, and this generally results in the inclusion of Catalent in our customers’ prescription product regulatory filings. Both of these factors frequently translate to long-duration supply relationships at an individual product level. We believe our customers value us because our depth of development solutions and advanced delivery technologies, intellectual property, consistent and reliable supply, geographic reach, and substantial expertise enable us to create a broad range of business and product solutions that can be customized to fit their individual needs. Today we employ more than 2,400 scientists and technicians and hold nearly 1,300 patents and patent applications in advanced delivery, drug and biologics formulation, and manufacturing. The aim of our offerings is to allow our customers to bring more products to market faster, and to develop and market differentiated new products that improve patient outcomes. We believe our leading market position and diversity of customers, offerings, regulatory categories, products, and geographies reduce our exposure to potential strategic and product shifts within the industry. We provide a number of proprietary, differentiated technologies, products, and service offerings to our customers across our advanced delivery technologies and development solutions platforms. The core technologies within our advanced delivery technologies platform include softgel capsules, our Zydis orally dissolving tablets, blow-fill-seal unit-dose liquids, adenoassociated virus (“AAV”) vectors, and a range of other oral, injectable, and respiratory delivery technologies.
The technologies and service offerings within our development solutions platform span the drug development process, ranging from our OptiForm Solution Suite for enhancement of bioavailability and other characteristics of early-stage molecules, and Gene Product Expression (“GPEx”), GPEx Boost, and SMARTag platforms for development of biologics and antibody-drug conjugates (“ADCs”), to formulation, analytical services, early-stage clinical development, and clinical trials supply, including our unique FlexDirect direct-to-patient and FastChain demand-led clinical supply solutions. Our offerings serve a critical need in the development and manufacturing of difficult-to-formulate products across a number of product types. We have advanced our technologies and grown our service offerings over more than 85 years through internal development, strategic alliances, in-licensing, and acquisitions. We initially introduced our softgel capsule technology in the 1930s and have continued to expand our range of new, technologically enhanced offerings. Since fiscal 2013, we have launched OptiShell, OptiMelt, Zydis Nano, Zydis Bio, OptiPact, the OptiForm Solution Suite, and our FlexDirect, SupplyFlex, and FastChain demand-led clinical supply solutions, as well as both clinical- and commercial-scale spray-drying formulation and 6 delivery services.
Our customers use these solutions to advance their products, including the first commercially marketed product using our OptiShell non-gelatin soft capsule technology. We have also augmented our portfolio through acquisitions. In fiscal 2017, we expanded our early development capabilities through the September 2016 acquisition of Pharmatek Laboratories, Inc. (“Pharmatek”), and we expanded our softgel development and manufacturing network through the February 2017 acquisition of Accucaps Industries Limited (“Accucaps”). In fiscal 2018, we acquired Cook Pharmica LLC (now known as Catalent Indiana, LLC (“Catalent Indiana”)) in order to enhance our biologics capabilities. In fiscal 2019, we acquired Juniper Pharmaceuticals, Inc. (“Juniper”), extending to the E.U. our early development capabilities, and Paragon Bioservices, Inc. (now known as Catalent Maryland, Inc. (“Catalent Maryland”)), adding advanced gene therapy development and manufacturing capabilities to our biologics business. In fiscal 2020, we augmented our gene therapy capabilities with the acquisition of certain assets, including two advanced biologics clinical development and manufacturing sites in southern Maryland, from Novavax, Inc. (“Novavax”) in July 2019; in January 2020, we acquired Bristol-Myers Squibb Company’s oral solid, biologics, and sterile product manufacturing and packaging facility in Anagni, Italy (“Anagni”); and, in February 2020, we acquired Masthercell Global Inc. (“MaSTherCell”), which develops and manufactures autologous and allogeneic cell therapies for third parties and provides a variety of related analytical services. In May 2020, we agreed to acquire a clinical packaging facility located in Shiga, Japan from Takeda-Teva Pharmaceuticals, Inc. and completed the acquisition on July 1, 2020. In large part due to our recent acquisitions and their subsequent organic growth, revenue contributions from our biologics business have grown from approximately 10% in fiscal 2014 to 33% in fiscal 2020. We believe our own internal innovation, supplemented by current and future external partnerships and acquisitions, will continue to strengthen and extend our leadership positions in the delivery and development of drugs, biologics, cell and gene therapies, and consumer health products.
Catalent employs over 11,000 people, including over 1,400 scientists, at more than 30 facilities across five continents, and in fiscal 2017 generated over $2 billion in annual revenue.
We believe that, through our investments in growth-enabling capacity and capabilities, our ongoing focus on operational and quality excellence, the sales of existing and introduction of new customer products, our innovation activities and patents, and our entry into new markets, we will continue to benefit from attractive and differentiated margins and realize the growth potential from these areas. We continue to invest in our sales and marketing activities, leading to growth in the number of active development programs for our customers. This has further enhanced our extensive, long-duration relationships and long-term contracts with a broad and diverse range of industry-leading customers. In fiscal 2020, we conducted business with 86 of the top 100 branded drug marketers, 24 of the top 25 generics marketers, 23 of the top 25 biologics marketers, and 22 of the top 25 consumer health marketers globally. Selected key customers include Pfizer, Johnson & Johnson, GlaxoSmithKline, Novartis, Roche, and Teva.
We have many long-standing relationships with our customers, particularly in advanced delivery technologies, where we tend to follow a prescription molecule through its lifecycle, from the development and launch of the original brand prescription, to potential generics or an over-the-counter switch. A prescription pharmaceutical product relationship with an innovator will often last many years, in several cases, two decades or more, extending from pre-clinical development through the end of the product’s life cycle. We serve customers who require innovative product development, superior quality, advanced manufacturing, and skilled technical services to support their development and marketed product needs. Our broad and diverse range of technologies closely integrates with our customers’ molecules and other active ingredients to yield final formulations and dose forms, and this generally results in the inclusion of Catalent in our customers’ prescription product regulatory filings. Both of these factors frequently translate to long-duration supply relationships at an individual product level. We believe our customers value us because our depth of development solutions and advanced delivery technologies, intellectual property, consistent and reliable supply, geographic reach, and substantial expertise enable us to create a broad range of business and product solutions that can be customized to fit their individual needs. Today we employ more than 2,400 scientists and technicians and hold nearly 1,300 patents and patent applications in advanced delivery, drug and biologics formulation, and manufacturing. The aim of our offerings is to allow our customers to bring more products to market faster, and to develop and market differentiated new products that improve patient outcomes. We believe our leading market position and diversity of customers, offerings, regulatory categories, products, and geographies reduce our exposure to potential strategic and product shifts within the industry. We provide a number of proprietary, differentiated technologies, products, and service offerings to our customers across our advanced delivery technologies and development solutions platforms. The core technologies within our advanced delivery technologies platform include softgel capsules, our Zydis orally dissolving tablets, blow-fill-seal unit-dose liquids, adenoassociated virus (“AAV”) vectors, and a range of other oral, injectable, and respiratory delivery technologies.
The technologies and service offerings within our development solutions platform span the drug development process, ranging from our OptiForm Solution Suite for enhancement of bioavailability and other characteristics of early-stage molecules, and Gene Product Expression (“GPEx”), GPEx Boost, and SMARTag platforms for development of biologics and antibody-drug conjugates (“ADCs”), to formulation, analytical services, early-stage clinical development, and clinical trials supply, including our unique FlexDirect direct-to-patient and FastChain demand-led clinical supply solutions. Our offerings serve a critical need in the development and manufacturing of difficult-to-formulate products across a number of product types. We have advanced our technologies and grown our service offerings over more than 85 years through internal development, strategic alliances, in-licensing, and acquisitions. We initially introduced our softgel capsule technology in the 1930s and have continued to expand our range of new, technologically enhanced offerings. Since fiscal 2013, we have launched OptiShell, OptiMelt, Zydis Nano, Zydis Bio, OptiPact, the OptiForm Solution Suite, and our FlexDirect, SupplyFlex, and FastChain demand-led clinical supply solutions, as well as both clinical- and commercial-scale spray-drying formulation and 6 delivery services.
Our customers use these solutions to advance their products, including the first commercially marketed product using our OptiShell non-gelatin soft capsule technology. We have also augmented our portfolio through acquisitions. In fiscal 2017, we expanded our early development capabilities through the September 2016 acquisition of Pharmatek Laboratories, Inc. (“Pharmatek”), and we expanded our softgel development and manufacturing network through the February 2017 acquisition of Accucaps Industries Limited (“Accucaps”). In fiscal 2018, we acquired Cook Pharmica LLC (now known as Catalent Indiana, LLC (“Catalent Indiana”)) in order to enhance our biologics capabilities. In fiscal 2019, we acquired Juniper Pharmaceuticals, Inc. (“Juniper”), extending to the E.U. our early development capabilities, and Paragon Bioservices, Inc. (now known as Catalent Maryland, Inc. (“Catalent Maryland”)), adding advanced gene therapy development and manufacturing capabilities to our biologics business. In fiscal 2020, we augmented our gene therapy capabilities with the acquisition of certain assets, including two advanced biologics clinical development and manufacturing sites in southern Maryland, from Novavax, Inc. (“Novavax”) in July 2019; in January 2020, we acquired Bristol-Myers Squibb Company’s oral solid, biologics, and sterile product manufacturing and packaging facility in Anagni, Italy (“Anagni”); and, in February 2020, we acquired Masthercell Global Inc. (“MaSTherCell”), which develops and manufactures autologous and allogeneic cell therapies for third parties and provides a variety of related analytical services. In May 2020, we agreed to acquire a clinical packaging facility located in Shiga, Japan from Takeda-Teva Pharmaceuticals, Inc. and completed the acquisition on July 1, 2020. In large part due to our recent acquisitions and their subsequent organic growth, revenue contributions from our biologics business have grown from approximately 10% in fiscal 2014 to 33% in fiscal 2020. We believe our own internal innovation, supplemented by current and future external partnerships and acquisitions, will continue to strengthen and extend our leadership positions in the delivery and development of drugs, biologics, cell and gene therapies, and consumer health products.
Operating Segments of Catalent
In fiscal 2020, we engaged in a business reorganization of our operating segments to better align our internal business unit structure with our “Follow the Molecule” strategy and the increased focus on our biologics-related offerings. Under the revised structure, we changed the components and names of three of our four operating segments (with our fourth segment, Clinical Supply Services, unchanged). We currently operate in four operating segments, which also constitute our four reporting segments: Softgel and Oral Technologies, Biologics, Oral and Specialty Delivery, and Clinical Supply Services,
Softgel and Oral Technologies
Through our Softgel and Oral Technologies segment, we provide formulation, development, and manufacturing services for soft capsules, or “softgels,” as well as large-scale manufacturing of oral solid dose forms for pharmaceutical and consumer health markets, and supporting ancillary services.
Biologics
Our Biologics segment provides biologic cell-line, cell therapy and viral vector gene therapy development and manufacturing; formulation, development, and manufacturing for parenteral dose forms, including prefilled syringes, vials, and cartridges; and analytical development and testing services for large molecules. The business has extensive expertise in development, scale up, and commercial manufacturing. Representative customers of Biologics include Eli Lilly, Teva, Mylan, Roche, AstraZeneca, Novartis, Sarepta, and Genentech, along with multiple innovative small and mid-tier pharmaceutical and biologics customers
Oral and Speciality Delivery
Our Oral and Specialty Delivery segment provides advanced formulation development and manufacturing across a range of technologies along with integrated downstream clinical development and commercial supply solutions. The technologies cover a broad range of oral (including our proprietary Zydis orally dissolving tablets and many bioavailability enhancement (BAE) technologies for both immediate and controlled-release tablets and capsules), respiratory and inhaled dose forms, including blow-fill-seal, metered dose inhalers, dry powder inhalers, and nasal applicators. Representative customers of Oral and Specialty Delivery include Johnson & Johnson, Pfizer, Bayer, Perrigo, Roche, and Genentech but also many small and midsized companies involved in the clinical development space.
Critical Supply Services
Our Clinical Supply Services segment provides manufacturing, packaging, storage, distribution, and inventory management for drugs and biologics in clinical trials. We offer customers flexible solutions for clinical supplies production and provide distribution and inventory management support for both simple and complex clinical trials. This includes overencapsulation where needed; supplying placebos, comparator drug procurement, and clinical packages and kits for physicians and patients; inventory management; investigator kit ordering and fulfillment; and return supply reconciliation and reporting. We support trials in all regions of the world through our facilities and distribution network. In recent years, we have continued to expand and extend our network, with significant expansions in Kansas City, Missouri and Singapore and new facilities in China and, effective July 2020, in Japan. We are the leading provider of integrated development solutions and one of the leading providers of clinical trial supplies. Representative customers of Clinical Supply Services include Merck KGaA, IQVIA, Eli Lilly, AbbVie, and Incyte Corporation
Softgel and Oral Technologies
Through our Softgel and Oral Technologies segment, we provide formulation, development, and manufacturing services for soft capsules, or “softgels,” as well as large-scale manufacturing of oral solid dose forms for pharmaceutical and consumer health markets, and supporting ancillary services.
Biologics
Our Biologics segment provides biologic cell-line, cell therapy and viral vector gene therapy development and manufacturing; formulation, development, and manufacturing for parenteral dose forms, including prefilled syringes, vials, and cartridges; and analytical development and testing services for large molecules. The business has extensive expertise in development, scale up, and commercial manufacturing. Representative customers of Biologics include Eli Lilly, Teva, Mylan, Roche, AstraZeneca, Novartis, Sarepta, and Genentech, along with multiple innovative small and mid-tier pharmaceutical and biologics customers
Oral and Speciality Delivery
Our Oral and Specialty Delivery segment provides advanced formulation development and manufacturing across a range of technologies along with integrated downstream clinical development and commercial supply solutions. The technologies cover a broad range of oral (including our proprietary Zydis orally dissolving tablets and many bioavailability enhancement (BAE) technologies for both immediate and controlled-release tablets and capsules), respiratory and inhaled dose forms, including blow-fill-seal, metered dose inhalers, dry powder inhalers, and nasal applicators. Representative customers of Oral and Specialty Delivery include Johnson & Johnson, Pfizer, Bayer, Perrigo, Roche, and Genentech but also many small and midsized companies involved in the clinical development space.
Critical Supply Services
Our Clinical Supply Services segment provides manufacturing, packaging, storage, distribution, and inventory management for drugs and biologics in clinical trials. We offer customers flexible solutions for clinical supplies production and provide distribution and inventory management support for both simple and complex clinical trials. This includes overencapsulation where needed; supplying placebos, comparator drug procurement, and clinical packages and kits for physicians and patients; inventory management; investigator kit ordering and fulfillment; and return supply reconciliation and reporting. We support trials in all regions of the world through our facilities and distribution network. In recent years, we have continued to expand and extend our network, with significant expansions in Kansas City, Missouri and Singapore and new facilities in China and, effective July 2020, in Japan. We are the leading provider of integrated development solutions and one of the leading providers of clinical trial supplies. Representative customers of Clinical Supply Services include Merck KGaA, IQVIA, Eli Lilly, AbbVie, and Incyte Corporation
Sales and Marketing of Catalent
Our target customers include large pharmaceutical and biotechnology companies, mid-size, emerging, and specialty pharmaceutical and biotechnology companies, and consumer health companies, along with companies in other selected healthcare market segments such as animal health and medical devices, and companies in adjacent industries, such as cosmetics. We have longstanding, extensive relationships with leading pharmaceutical and biotechnology customers. In fiscal 2020, we did business with 86 of the top 100 branded drug marketers, 24 of the top 25 generics marketers, 23 of the top 25 biologics marketers, and 22 of the top 25 consumer health marketers globally, as well as with more than 1,000 other customers. Faced with access, pricing, and reimbursement pressures as well as other market challenges, large pharmaceutical and biotechnology companies have increasingly sought partners to enhance the clinical competitiveness of their drugs and biologics and improve the productivity of their research and development activities, while reducing their fixed cost base. Many mid-size, emerging, and specialty pharmaceutical and biotechnology companies, while facing the same pricing and market pressures, have chosen not to build a full infrastructure, but rather to partner with other companies through licensing agreements or outsourcing to access the critical skills, technologies, and services required to bring their products to market. Consumer health companies require rapidly developed, innovative dose forms and formulations to keep up with the fast-paced over-the-counter medication, vitamins, and personal care markets. These market segments are all important to our growth, but require distinct solutions, marketing and sales approaches, and market strategy.
We follow a hybrid demand-generation organization model, with strategic account teams offering the full breadth of Catalent’s solutions, and technical specialist teams providing the in-depth technical knowledge and practical experience essential for each individual offering. Our sales organization currently consists of more than 160 full-time, experienced sales professionals, supported by inside sales and sales operations. We also have built a dedicated strategic marketing team, providing strategic market and product planning and management for our offerings. As part of our marketing efforts, we participate in major trade shows relevant to our offerings globally and ensure adequate visibility to our offerings and solutions through a comprehensive print and on-line advertising and publicity program. We believe that Catalent is a strong brand with high overall awareness in our established markets and universe of target customers, and that our brand identity is a competitive advantage for us.
We follow a hybrid demand-generation organization model, with strategic account teams offering the full breadth of Catalent’s solutions, and technical specialist teams providing the in-depth technical knowledge and practical experience essential for each individual offering. Our sales organization currently consists of more than 160 full-time, experienced sales professionals, supported by inside sales and sales operations. We also have built a dedicated strategic marketing team, providing strategic market and product planning and management for our offerings. As part of our marketing efforts, we participate in major trade shows relevant to our offerings globally and ensure adequate visibility to our offerings and solutions through a comprehensive print and on-line advertising and publicity program. We believe that Catalent is a strong brand with high overall awareness in our established markets and universe of target customers, and that our brand identity is a competitive advantage for us.
Seasonality of Catalent's earnings
Our annual financial reporting periods end on June 30. Our revenue and net earnings are generally higher in the third and fourth quarters of each fiscal year, with our first fiscal quarter typically generating our lowest revenue of any quarter, and our last fiscal quarter typically generating our highest revenue. These fluctuations are primarily the result of the timing of our, and our customers’, annual operational maintenance periods at locations in continental Europe and the U.K., the seasonality associated with pharmaceutical and biotechnology budgetary spending decisions, clinical trial and research and development schedules, the timing of new product launches and length of time needed to obtain full market penetration, and, to a lesser extent, the time of the year some of our customers’ products are in higher demand.
Contractual Agreements of Catalent
We generally enter into a broad range of contractual arrangements with our customers, including agreements with respect to feasibility, development, supply, licenses, and quality. The terms of these contracts vary significantly depending on the offering and customer requirements. Some of our agreements may include a variety of revenue arrangements, such as fee-forservice, minimum volume commitments, royalties, manufacturing preparation services, profit-sharing, and fixed fees. We employ a range of capacity access approaches, from standard to completely dedicated capacity models, based on customer and product needs. We generally secure pricing and other contract mechanisms in our supply agreements to allow for periodic resetting of pricing terms, and, in some cases, these agreements permit us to raise or renegotiate pricing in the event of certain price increases for the raw materials we use to make products. Our typical supply agreements include indemnification from our customers for product liability and intellectual property matters and caps on our contractual liabilities, subject in each case to negotiated exclusions. The terms of our manufacturing supply agreements range from three to seven years with regular renewals of one to three years, although some of our agreements are terminable upon much shorter notice periods, such as 30 or 90 days. For our development solutions offerings, we may enter into master service agreements, which provide for standardized terms and conditions and make it easier and faster for customers with multiple development needs to access our offerings.
Backlog of Catalent's products
While we generally have long-term supply agreements that provide for a revenue stream over a period of years, our backlog represents, as of a point in time, future service revenues from work not yet completed. For our Softgel and Oral Technologies, Biologics, and Oral and Specialty Delivery segments, backlog represents firm orders for manufacturing services and includes minimum volumes, where applicable. For our Clinical Supply Services segment, backlog represents estimated future service revenues from work not yet completed under signed contracts. Using these methods of reporting backlog, as of June 30, 2020, our backlog was $2,282.3 million compared to $1,349.8 million as of June 30, 2019, including $424.7 million and $366.3 million, respectively, related to our Clinical Supply Services segment. We expect to recognize as revenue approximately 72% of the value of the backlog in existence as of June 30, 2020 by the completion of fiscal 2021. To the extent projects are delayed, the timing of our revenue could be affected. If a customer cancels an order, we may be reimbursed for the costs we have incurred. For orders that are placed inside a contractual firm period, we generally have a contractual right to payment in the event of cancellation. Fluctuations in our reported backlog levels also result from the timing and order pattern of our customers who often seek to manage their level of inventory on hand. Because of customer ordering patterns, our backlog reported for certain periods may fluctuate and may not be indicative of future results
Manufacturing Capabilities of Catalent
We operate manufacturing facilities, development centers, and sales offices throughout the world. As of June 30, 2020, we had 53 facilities (4 geographical locations operate as multiple facilities because they support more than one reporting segment) on four continents with 7.1 million square feet of manufacturing, laboratory, and related space. In addition, in May 2020, we agreed to acquire a clinical packaging facility in Shiga, Japan from Takeda-Teva Pharmaceuticals, Inc.. The acquisition closed in July 2020. Our manufacturing capabilities include the full suite of competencies relevant to support each site’s activities, including regulatory, quality assurance, and in-house validation. We operate our manufacturing facilities and development centers in accordance with cGMP or other applicable requirements. All of these sites are registered where required with the FDA or other applicable regulatory agencies, such as the EMA. In some cases, our sites are registered with multiple regulatory agencies. We have invested $860.4 million in our manufacturing and development facilities since fiscal 2018 for improvements and expansions, including $465.8 million for capital expenditures in fiscal 2020. We believe that our sites and equipment are in good condition, are well maintained, and are able to operate at or above present levels for the foreseeable future, in all material respects. Our manufacturing operations are focused on employee health and safety, regulatory compliance, operational excellence, continuous improvement, and process standardization across the organization. In fiscal 2020, we achieved approximately 97% on-time shipment delivery versus customer request date across our network as a result of this focus. Our manufacturing operations are structured around an enterprise management philosophy and methodology that utilizes principles and tools common to a number of quality management programs, including Lean Six Sigma and Lean Manufacturing.
Raw Materials used by Catalent
We use a broad and diverse range of raw materials in the design, development, and manufacture of our products. This includes, but is not limited to, key materials such as gelatin, starch, and iota carrageenan for our Softgel and Oral Technologies segment; packaging films for our Clinical Supply Services segment; and glass vials and syringes for injectable fill-finish in our Biologics segment, and resin for our blow-fill-seal business in our Oral and Specialty Delivery segment. The raw materials that we use are sourced externally on a global basis. Globally, our supplier relationships could be interrupted due to natural disasters and international supply disruptions, including those caused by pandemics or geopolitical and other issues. For example, commercially usable gelatin is available from a limited number of sources. In addition, much of the gelatin we use is bovinederived. Past concerns of contamination from Bovine Spongiform Encephalopathy (“BSE”) have narrowed the number of possible sources of particular types of gelatin. If there were a future disruption in the supply of gelatin from any one or more key suppliers, there can be no assurance that we could obtain an alternative supply from our other suppliers. Any future restriction that were to emerge on the use of bovine-derived gelatin from certain geographic sources due to concerns of contamination from BSE could hinder our ability to timely supply our customers with products and the use of alternative nonbovine-derived gelatin for specific customer products could be subject to lengthy formulation, testing and regulatory approval periods. We work very closely with our suppliers to assure continuity of supply while maintaining excellence in material quality and reliability. We continually evaluate alternate sources of supply, although we do not frequently pursue regulatory qualification of alternative sources for key raw materials due to the strength of our existing supplier relationships, the reliability of our current supplier base, and the time and expense associated with the regulatory process. Although a change in suppliers could require significant effort or investment by us in circumstances where the items supplied are integral to the performance of our products or incorporate specialized material such as gelatin, we do not believe that the loss of any existing supply arrangement would have a material adverse effect on our business
Competition of Catalent
We compete with multiple companies as to each of our offerings and in every region of the globe in which we operate, including with other companies that offer conventional and advanced delivery technologies, clinical trials support, outsourced dose form, biologics or cell and gene therapy manufacturing, or development services to pharmaceutical, biotechnology, and consumer health companies based in North America, South America, Europe, and the Asia-Pacific region. We also compete in some cases with the internal operations of those pharmaceutical, biotechnology, and consumer health customers that also have manufacturing capabilities and choose to source these services internally. Some of our competitors are substantially larger than we are and have access to more substantial resources, which could be deployed to expand their range of offerings or capacity. 16 Competition is driven by proprietary technologies and know-how (where relevant), capabilities, consistency of operational performance, quality, price, value, responsiveness, and speed. While we do have competitors that compete with us in our individual offerings, and a few competitors that compete across many of our offerings, we do not believe we have competition from any directly comparable company.
Employees of Catalent
As of June 30, 2020, we had more than 13,900 employees in 53 facilities on4continents: 22 facilities are in the United States, with certain employees at one facility being represented by a labor organization with their terms and conditions of employment being subject toacollective bargaining agreement. National works councils and/or labor organizations are active at all seventeen of our European facilities consistent with labor environments and laws in European countries. Similar relationships with labor organizations or national works councils exist at our plants in Argentina, Brazil, and Canada. Our management believes that our employee relations are satisfactory.
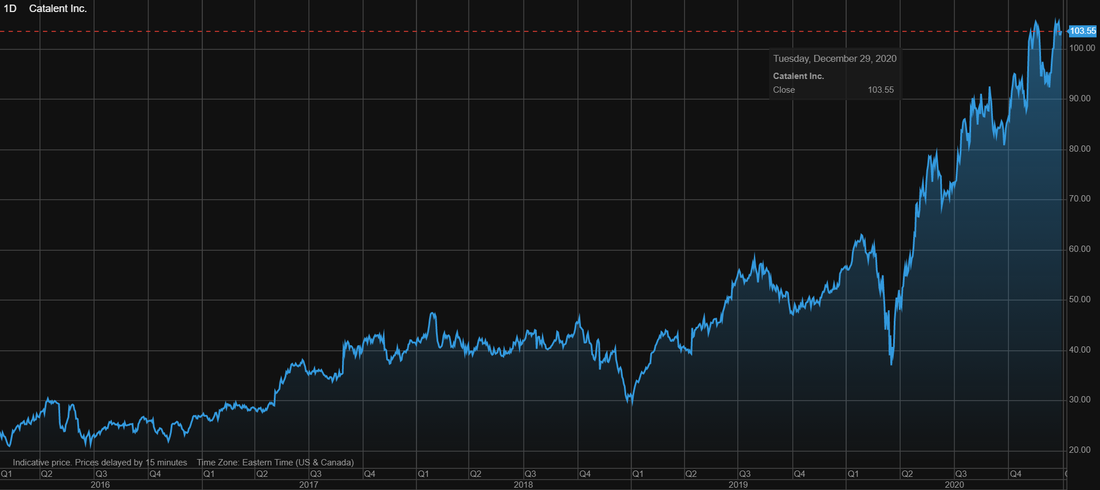
Catalent (CTLT) stock price history over the last 5 years
The image below shows the stock price history of Catalent (CTLT). Over the last 5 years the stock of Catalent has increased significantly by 340.7%. Returns no investor will say no to.